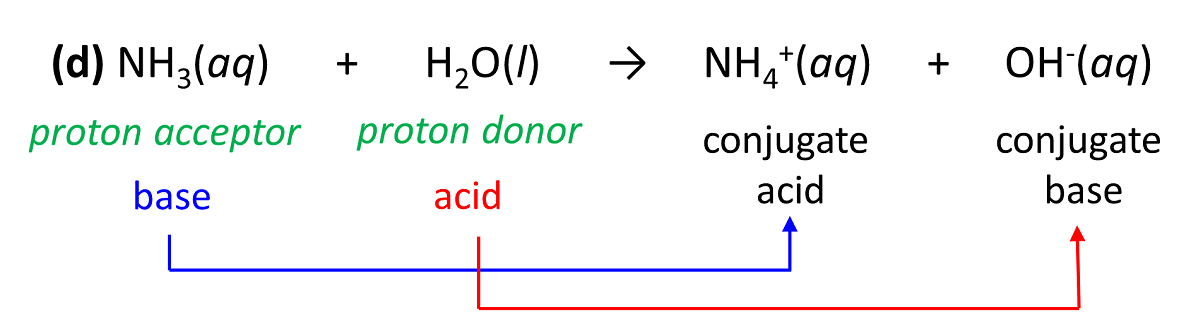
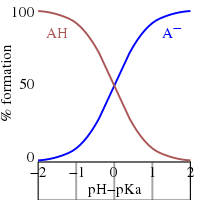
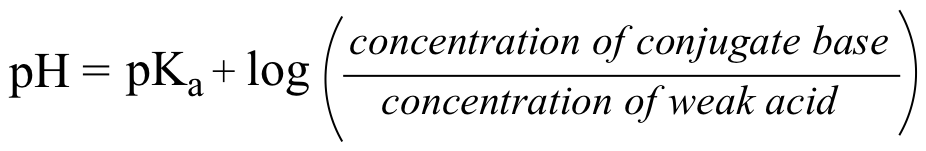How to Identify the Major Species in a Mixture of Weak and Strong Acids or Bases | Chemistry | Study.com
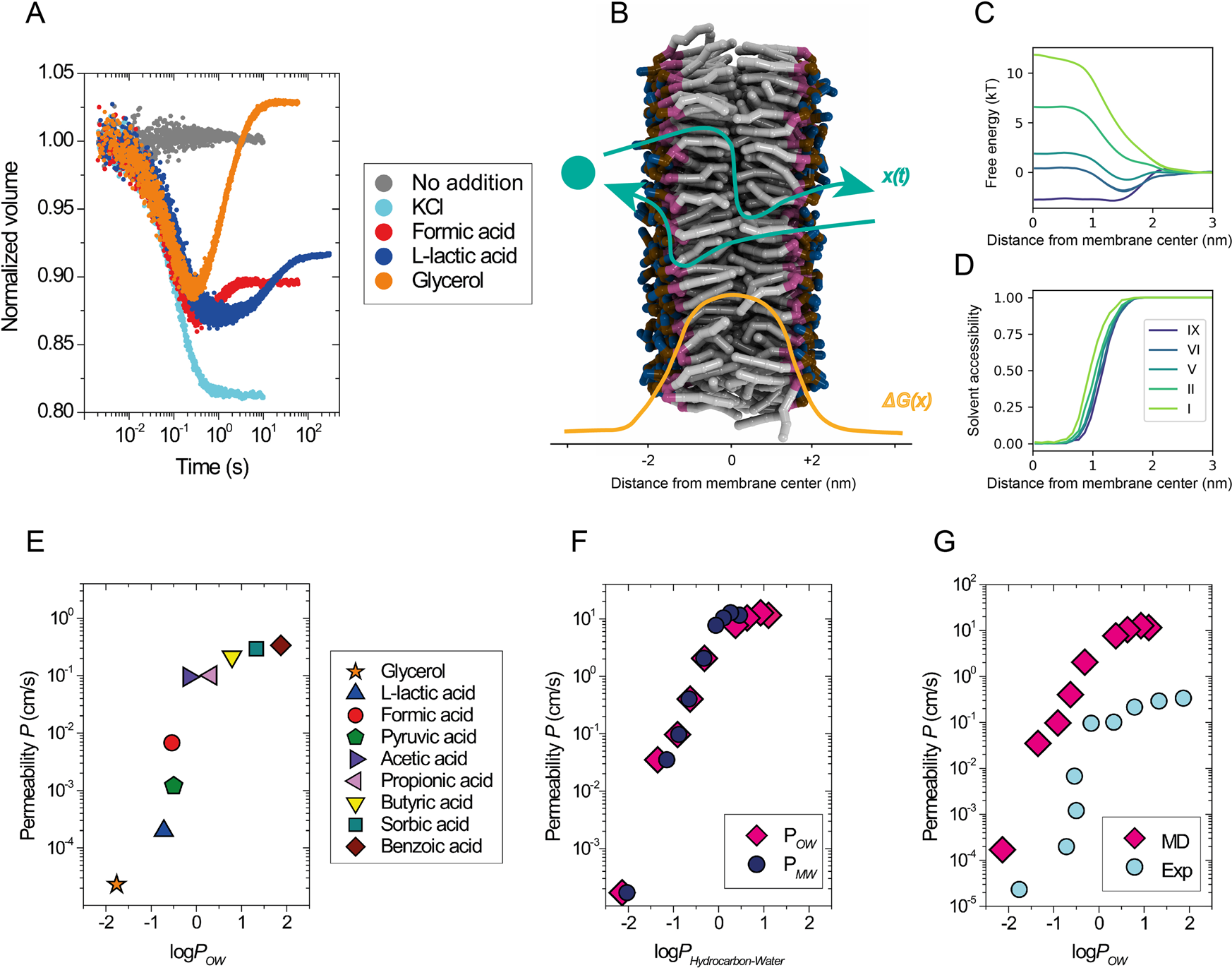
Strong Relationships in Acid-Base Chemistry – Modeling Protons Based on Predictable Concentrations of Strong Ions, Total Weak Acid Concentrations, and pCO2 | PLOS ONE
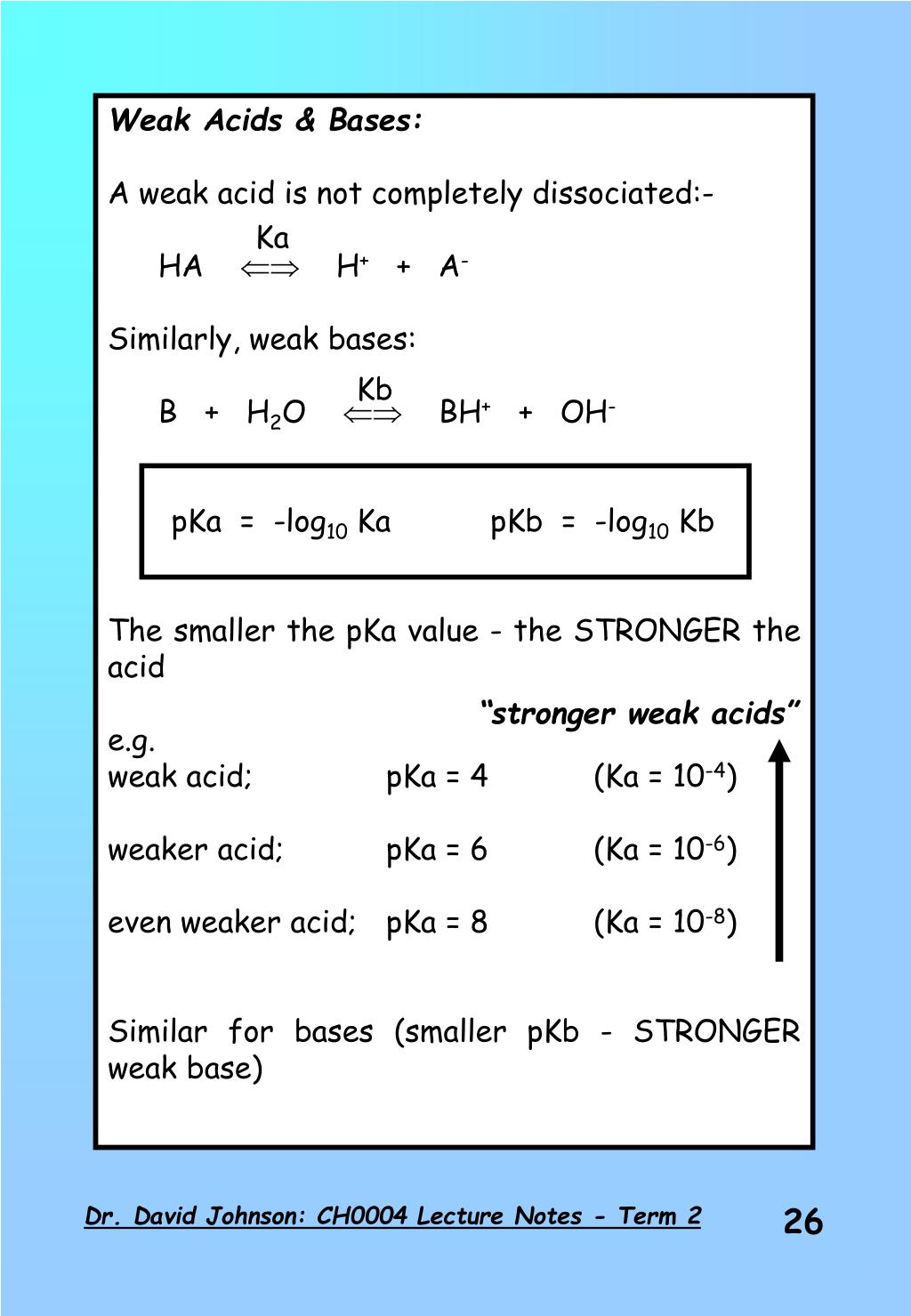
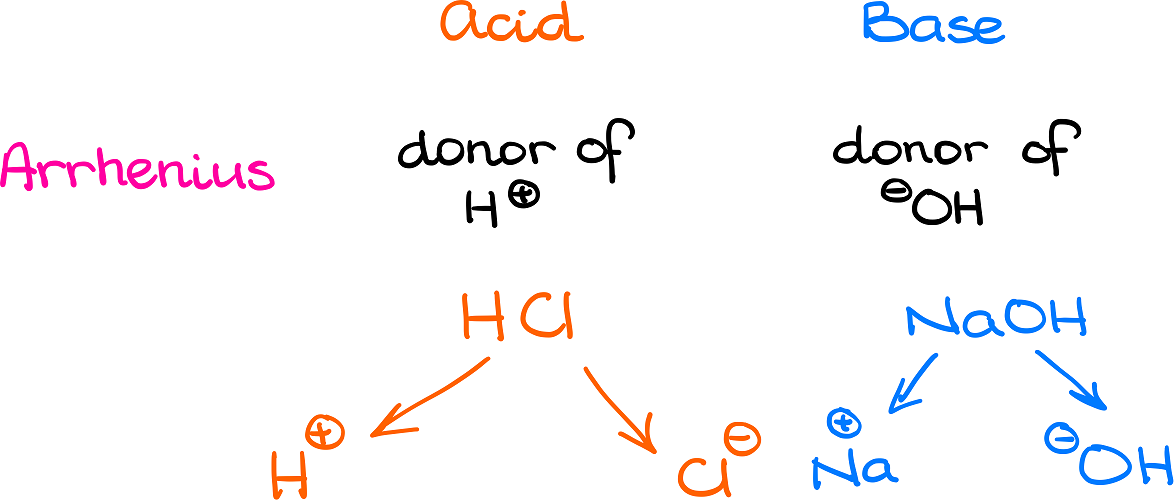
PPT - Weak Acids & Bases: A weak acid is not completely dissociated:- HA H + + A - PowerPoint Presentation - ID:3428717

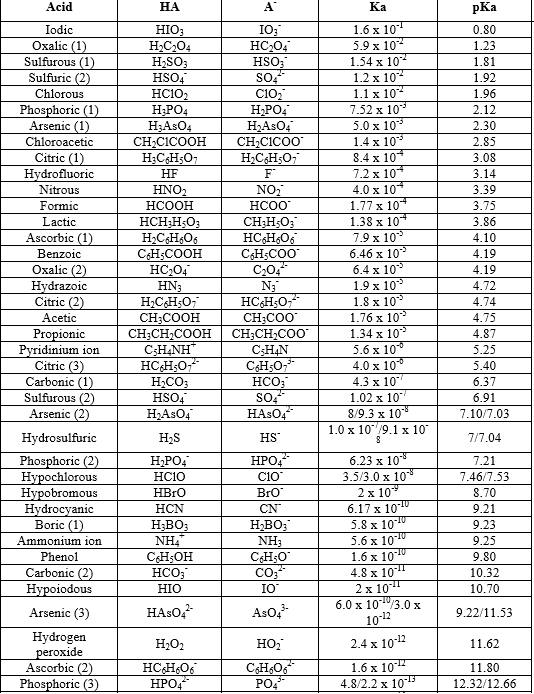
It has been found that the pH of a 0.01 M solution of an organic acid is 4.15 . Calculate the concentration of the anion, the ionization constant of the acid and its pKa .

Recent trends on density functional theory–assisted calculations of structures and properties of metal–organic frameworks and metal–organic frameworks-derived nanocarbons | Journal of Materials Research | Cambridge Core

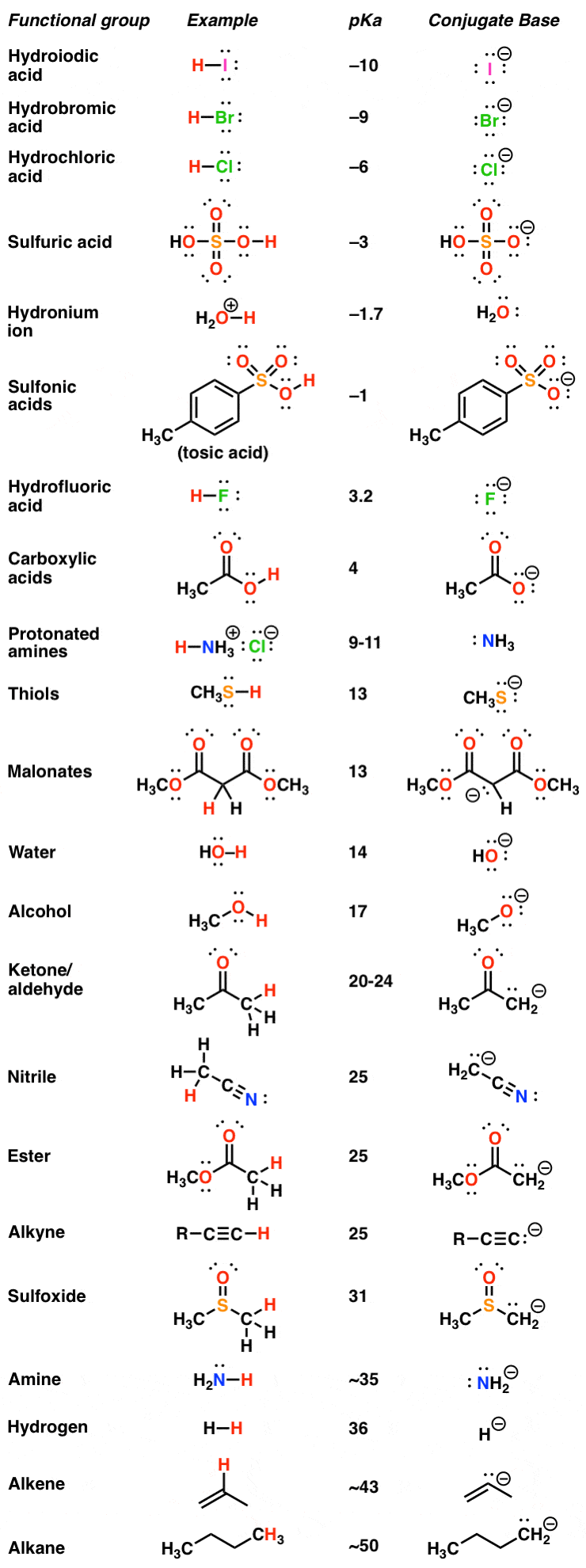
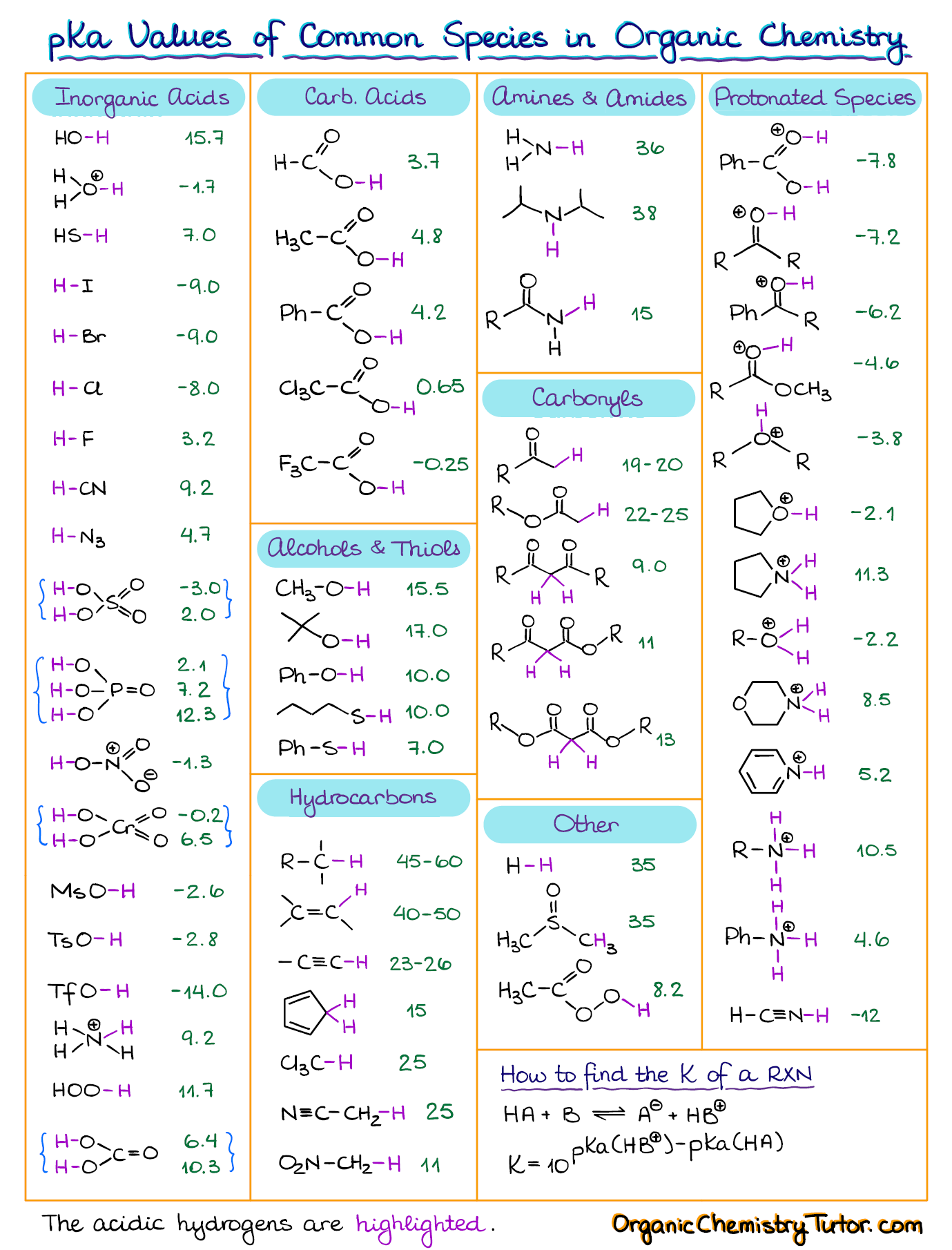
3.3: pKa of Organic Acids and Application of pKa to Predict Acid-Base Reaction Outcome - Chemistry LibreTexts