HIZENTRA 200 MG/ML ROZTOK NA SUBKUTÁNNU INJEKCIU sol icu 1x50 ml/10 g (liek.inj.skl.) - Príbalový leták

PDF) Molecular properties of human IgG subclasses and their implications for designing therapeutic monoclonal antibodies against infectious diseases
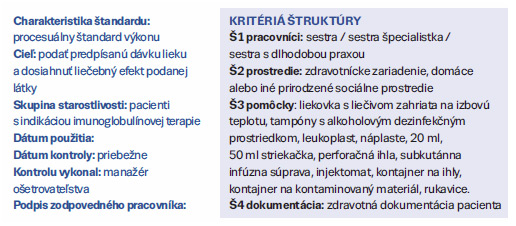
Návrh ošetrovateľského štandardu aplikácie imunoglobulínového prípravku subkutánnou cestou (číslo 3 / 2018) | Archiv | Odborné články | FLORENCE - Odborný časopis pro ošetřovatelství a ostatní zdravotnické profese

The FcRn inhibitor rozanolixizumab reduces human serum IgG concentration: A randomized phase 1 study | Science Translational Medicine
Nima Rezaei Asghar Aghamohammadi Luigi D. Notarangelo Editors Definition, Diagnosis, and Management Second Edition
FINAL REPORT NOVEMBER 22,2019 PRESENTED BY: CANADIAN IMMUNODEFICIENCIES PATIENT ORGANIZATION SCIG SHORTAGE REPORT

PDF) Clinical Predictors of Relapse in a Cohort of Steroid-Treated Patients With Well-Controlled Myasthenia Gravis

Molecular properties of human IgG subclasses and their implications for designing therapeutic monoclonal antibodies against infectious diseases - ScienceDirect

The FcRn inhibitor rozanolixizumab reduces human serum IgG concentration: A randomized phase 1 study | Science Translational Medicine

Molecular properties of human IgG subclasses and their implications for designing therapeutic monoclonal antibodies against infectious diseases - ScienceDirect