Question Video: Calculating the Moles of a Gas in a Given Volume by Determining the Molar Gas Volume | Nagwa

Question Video: Determining the Relationship between Temperature and Number of Moles of an Ideal Gas | Nagwa

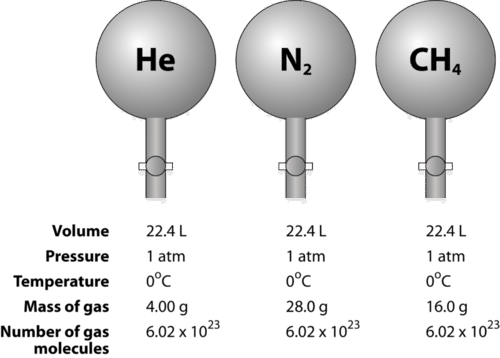
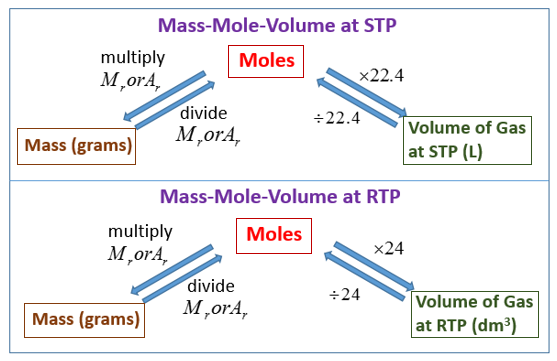
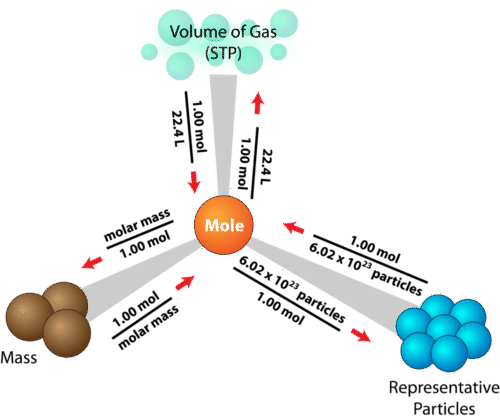
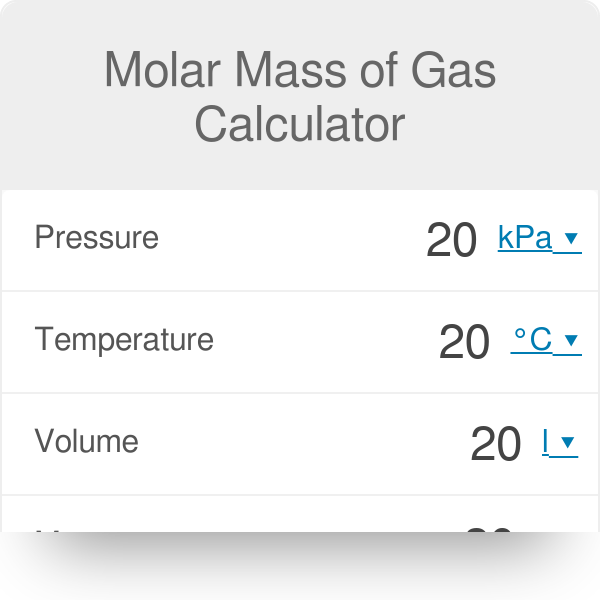
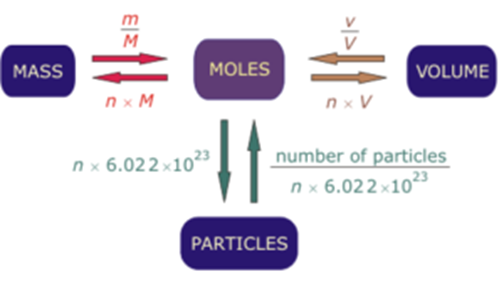
molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises

SOLVED: Calculate the theoretical number of moles of hydrogen gas produced: What would be the effect on the molar volume if oxygen instead of hydrogen gas (as in equation below) had been